Last Update 07 Apr 26
KYMR: Future Type 2 Disease Data Will Drive Oral Degrader Upside
Analysts' blended price target for Kymera Therapeutics has shifted modestly within a range of about $100 to $140 as they weigh updated Q4 data, progress across the degrader pipeline, and the potential of the STAT6 program as an oral alternative to existing biologics in type 2 diseases.
Analyst Commentary
Recent research notes cluster around a constructive stance on Kymera Therapeutics, with most firms adjusting price targets within a relatively tight US$100 to US$140 band as they reassess Q4 updates, the degrader pipeline, and the potential of the STAT6 program in type 2 diseases.
Bullish Takeaways
- Bullish analysts highlight the STAT6 degrader KT-621 as a key value driver, framing it as a potential oral alternative to existing biologics in type 2 diseases and pointing to supportive preclinical, Phase 1 healthy volunteer, and early patient data.
- Some research notes argue Kymera could become a category leader in oral protein degraders, which they view as central to the long term growth story and as a reason to assign higher price targets within the current US$100 to US$140 range.
- Analysts who are raising targets point to expanding clinical proof around the degrader platform and suggest that oral differentiation versus injectable biologics could support broader adoption if upcoming data stays consistent with early signals.
- One major house describes the risk reward profile as attractive at recent levels, citing Kymera’s first mover position in immunology degraders and the breadth of the opportunity set in type 2 diseases and related indications.
Bearish Takeaways
- Bearish analysts, or those trimming price targets, reflect caution around execution risk, especially the need to convert encouraging early stage readouts into larger, well controlled trials that can support long term revenue and cash flow visibility.
- The focus on a growing type 2 disease market is recognized, but some research points stress that competition from existing advanced therapies and future entrants could pressure pricing power and eventual market share assumptions.
- There is an undercurrent of concern that the investment narrative into 2026 remains largely unchanged after Q4, which can limit near term re rating potential unless upcoming clinical or pipeline milestones materially shift expectations.
- A few target cuts within the group underscore sensitivity to pipeline timing, regulatory risk, and the need for continued clean safety and efficacy data, all of which feed into a more conservative stance on how quickly Kymera can realize its full degrader opportunity.
What's in the News
- Positive BroADen Phase 1b atopic dermatitis data for KT-621 featured in a late breaking oral presentation at the American Academy of Dermatology Annual Meeting, with deep STAT6 degradation in skin and blood, reductions in Type 2 inflammatory biomarkers, and encouraging clinical activity alongside a favorable safety profile (company announcement).
- Initiation of the BREADTH Phase 2b global, randomized, double blind, placebo controlled trial of KT-621 in approximately 264 adults with moderate to severe eosinophilic asthma. The study includes three oral dose levels tested over 12 weeks, with plans to use the results to inform future Phase 3 studies across multiple Type 2 indications (company announcement).
- Ongoing BROADEN2 Phase 2b trial of KT-621 in moderate to severe atopic dermatitis in adolescent and adult patients, positioned alongside the asthma study to support broader KT-621 development across Type 2 diseases (company announcement).
- Company outlines broader 2026 preclinical and clinical plans for its oral immunology pipeline, including KT-621 progress and partnered IRAK4 degrader KT-485/SAR447971 with Sanofi, with a Phase 1 clinical trial for the partnered program expected to begin in 2026 (company announcement).
- Kymera files a US$500m at the market follow on equity offering of common stock, providing an additional potential source of capital for ongoing R&D and corporate activities (company filing).
- Board approves amendments to the Second Amended and Restated Bylaws to define exclusive forum provisions for certain corporate and securities law claims. The changes align the company’s governance language with recent Delaware law changes and specify federal district courts for Securities Act and Exchange Act related actions (company filing).
Valuation Changes
- Fair Value: Modelled fair value stays unchanged at about $119.14 per share, indicating no shift in this central estimate.
- Discount Rate: The discount rate edges up slightly from 7.09% to about 7.09%, a very small adjustment in the required return assumption.
- Revenue Growth: The long term revenue growth assumption remains effectively flat, moving fractionally from a 0.96% decline to about a 0.96% decline.
- Net Profit Margin: The assumed net profit margin increases from about 12.87% to roughly 14.76%, indicating a more optimistic view on future profitability at scale.
- Future P/E: The future P/E multiple comes down from about 2,984x to around 2,603x, still a very large ratio that shows how sensitive outcomes are to long term earnings assumptions.
Key Takeaways
- Advancing clinical programs and strategic partnerships could increase market share and positively impact future revenue and earnings.
- Solid cash runway supports focused R&D investments, potentially boosting long-term growth without immediate financing pressures.
- High R&D expenses and reliance on partnerships pose risks to Kymera's long-term financial health and ability to maintain a leadership position in their sector.
Catalysts
About Kymera Therapeutics- Together with its subsidiary, a clinical-stage biopharmaceutical company, focuses on discovering and developing small molecule therapeutics that selectively degrade disease-causing proteins by harnessing the body’s own natural protein degradation system.
- Kymera Therapeutics plans to advance its STAT6 and TYK2 programs into several clinical stages, which could potentially increase future revenue due to the expansion into new treatment markets and therapeutic areas.
- The anticipated Phase II and III trials for their immunology pipeline aim to deliver biologics-like efficacy in oral form, which could enhance net margins by reducing manufacturing costs associated with biologics and potentially capturing a larger market share.
- The collaboration with Sanofi on the IRAK4 program, with expanded Phase II trials, positions Kymera to fast-track toward pivotal trials, potentially accelerating time-to-market and impacting future earnings positively.
- The company's strategy to introduce at least one new IND per year could expand their pipeline steadily, offering opportunities for revenue growth from licensing deals or partnerships.
- With a significant cash runway extending into mid-2027, Kymera can support its R&D activities without immediate pressure for additional financing, allowing focused investment in high-potential programs that could drive long-term earnings growth.
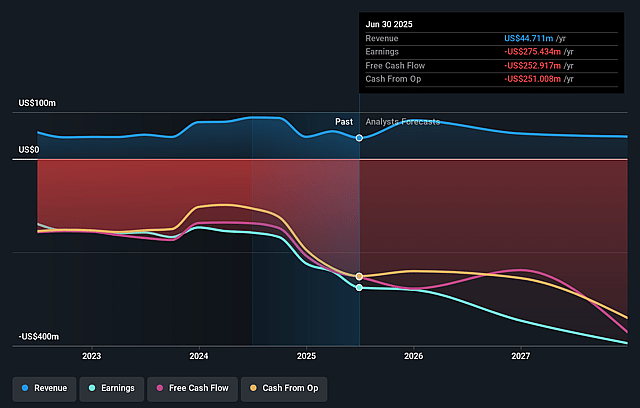
Kymera Therapeutics Future Earnings and Revenue Growth
Assumptions
How have these above catalysts been quantified?
- Analysts are assuming Kymera Therapeutics's revenue will remain fairly flat over the next 3 years.
- Analysts are not forecasting that Kymera Therapeutics will become profitable in next 3 years. To represent the Analyst Price Target as a Future PE Valuation we will estimate Kymera Therapeutics's profit margin will increase from -794.0% to the average US Biotechs industry of 14.8% in 3 years.
- If Kymera Therapeutics's profit margin were to converge on the industry average, you could expect earnings to reach $5.6 million (and earnings per share of $0.06) by about April 2029, up from -$311.4 million today.
- In order for the above numbers to justify the price target of the analysts, the company would need to trade at a PE ratio of 2603.7x on those 2029 earnings, up from -22.4x today. This future PE is greater than the current PE for the US Biotechs industry at 16.0x.
- Analysts expect the number of shares outstanding to grow by 7.0% per year for the next 3 years.
- To value all of this in today's terms, we will use a discount rate of 7.09%, as per the Simply Wall St company report.
Risks
What could happen that would invalidate this narrative?- The transition to a video format for financial updates may not significantly impact investor perception or the company’s market value, and does not directly address any operational or financial performance issues.
- Competition in the STAT6 space has increased, which may impact Kymera's ability to maintain its leadership position and could affect future revenue streams.
- Although significant progress is being made with partners like Sanofi, reliance on partnerships exposes Kymera to risks if partners face challenges in advancing clinical trials, potentially impacting future earnings.
- The financial performance shows high R&D expenses with $71.8 million spent in the fourth quarter alone, which could strain resources and impact net margins if new drugs don't reach successful commercialization.
- Despite a significant cash balance, the projected cash runway into mid-2027 suggests that sustained high operational costs could pose a risk to long-term financial health if projected clinical milestones or revenue targets are not met.
Valuation
How have all the factors above been brought together to estimate a fair value?
- The analysts have a consensus price target of $119.14 for Kymera Therapeutics based on their expectations of its future earnings growth, profit margins and other risk factors.
- However, there is a degree of disagreement amongst analysts, with the most bullish reporting a price target of $140.0, and the most bearish reporting a price target of just $91.0.
- In order for you to agree with the analysts, you'd need to believe that by 2029, revenues will be $38.1 million, earnings will come to $5.6 million, and it would be trading on a PE ratio of 2603.7x, assuming you use a discount rate of 7.1%.
- Given the current share price of $85.27, the analyst price target of $119.14 is 28.4% higher. Despite analysts expecting the underlying business to decline, they seem to believe it's more valuable than what the market thinks.
- We always encourage you to reach your own conclusions though. So sense check these analyst numbers against your own assumptions and expectations based on your understanding of the business and what you believe is probable.
Have other thoughts on Kymera Therapeutics?
Create your own narrative on this stock, and estimate its Fair Value using our Valuator tool.
Create NarrativeHow well do narratives help inform your perspective?
Disclaimer
AnalystConsensusTarget is a tool utilizing a Large Language Model (LLM) that ingests data on consensus price targets, forecasted revenue and earnings figures, as well as the transcripts of earnings calls to produce qualitative analysis. The narratives produced by AnalystConsensusTarget are general in nature and are based solely on analyst data and publicly-available material published by the respective companies. These scenarios are not indicative of the company's future performance and are exploratory in nature. Simply Wall St has no position in the company(s) mentioned. Simply Wall St may provide the securities issuer or related entities with website advertising services for a fee, on an arm's length basis. These relationships have no impact on the way we conduct our business, the content we host, or how our content is served to users. The price targets and estimates used are consensus data, and do not constitute a recommendation to buy or sell any stock, and they do not take account of your objectives, or your financial situation. Note that AnalystConsensusTarget's analysis may not factor in the latest price-sensitive company announcements or qualitative material.




